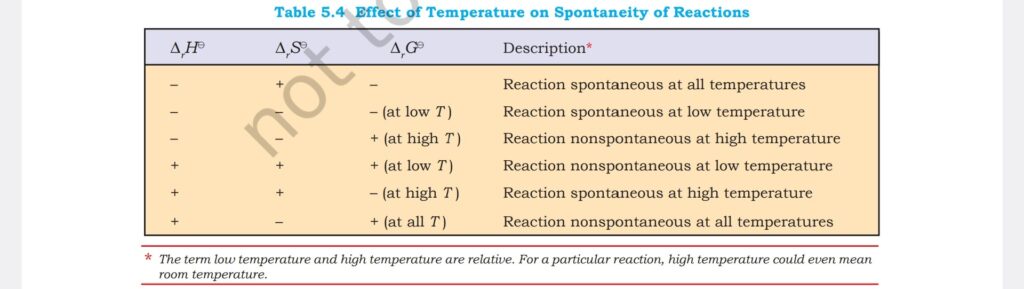
Class 11 formula



Number of spectral lines from n2 to n1 transition =(n2-n1)(n2-n1+1)/2



Classes of elements
Trends in modern periodic Table:
Periodic Trends in Properties of Elements-
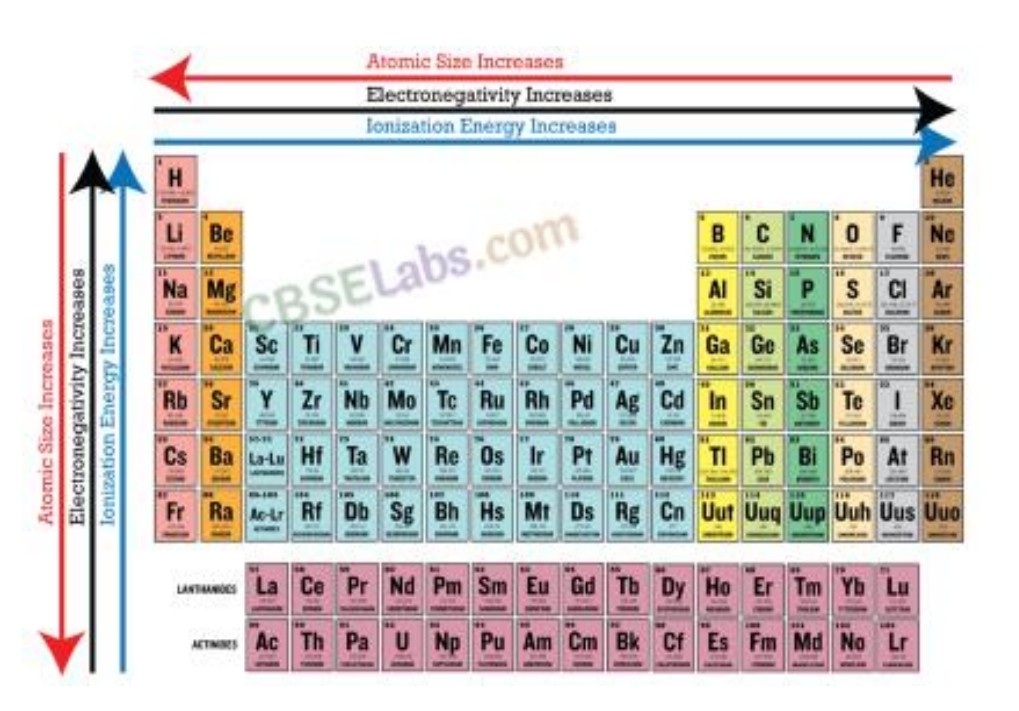
a) Atomic sizes- atomic radii decreases while going from left to right in a period and increases with atomic number in a group.
b) Ionisation enthalpies – increases across the periods and decreases down the group.
c) Electronegativity- increases across the periods and decreases down the group.
d) Electron gain enthalpy – becomes more negative across a period and less negative down a group.
e) Valency- the valence is either equal to the number of electron in the outermost orbit or eight minus this number.
f) Chemical reactivity – is highest at two extremes of a period and is lowest in the centre. Reactivity on the left extreme of a period is because of the ease of electron loss or low ionisation enthalpy.
g) Highly reactive elements do not exist in free state in nature, they usually exist in combined form.
h) Oxides – oxides form of the element on the left are basic and of the element on the right are acidic in nature. Oxides of elements in the centre are amphoteric or neutral.